
(Download) CAPF (AC) Exam, 2018 Paper "General Ability and Intelligence"
Exam Name: CAPF (AC)
Year: 2018
Subject: General Ability and Intelligence
1. The optical phenomenon that is responsible for the propagation of light signal through an optical fibre is
(a) interference
(b) scattering
(c) total internal reflection
(d) refraction
2. Which of the following statements about optical microscope is/are correct?
1. Both the eyepiece and objective of a microscope are convex lenses.
2. The magnification of a microscope increases with increase in focal length of the objective.
3. he magnification of a microscope depends upon the length of the microscope tube.
4. The eyepiece of a microscope is a concave lens.
Select the correct answer using the code given below.
(a) 1 and 3
(b) 3 only
(c) 3 and 4
(d) 1, 2 and 4
3. Which one of the following gemstones contains beryllium metal?
(a) Topaz
(b) Emerald
(c) Ruby
(d) Blue sapphire
4. Lowering the atmospheric pressure on a liquid
(a) increases the boiling point of the liquid
(b) lowers the boiling point of the liquid
(c) does not affect the boiling point of the liquid
(d) increases the time required for it to boil
5. The number of angular and radial nodes for 4d orbital is respectively
(a) 2 and 1
(b) 1 and 2
(c) 3 and 1
(d) 4 and 0
6. Which one of the following statements about proteins in mammalian cells is correct?
(a) Proteins in mammalian cells are synthesized in cell membrane.
(b) Proteins in mammalian cells are not synthesized in cell membrane but directly absorbed from food.
(c) Proteins in mammalian cells are synthesized in rough endoplasmic reticulum
(d) Proteins in mammalian cells are synthesized in Golgi apparatus.
7. Which one of the following statements about transport mechanism in
plants is correct?
(a) Diffusion of ions and small molecules across cell membrane requires
energy.
(b) Active transport of ions and small molecules across cell membrane against
concentration gradient requires energy.
(c) Cells conserve energy and no energy is required for transport of ions and
small molecules against concentration gradient.
(d) Cells do not transport ions across their membrane as these are charged
molecules.
8. If the amplitude of oscillation of a simple pendulum is very small,
then its time period of oscillation.
1. depends on the length of the pendulum, L
2. depends on the acceleration due to gravity g
3. depends upon the mass of the bob of the pendulum, m
4. does not depend upon the amplitude of the pendulum, A
Select the correct answer using the code given below.
(a) 1, 2 and 3
(b) 1, 2 and 4
(c) 2, 3 and 4
(d) 1 and 4 only
9. The term LASER stands for
(a) Light Amplification by Stimulated Emission of Radiation
(b) Light Amplification by Spontaneous Emission of Radiation
(c) Light Amplification by Stimulated Emission of Rays
(d) Light Amplification by Stimulated Energy of Radiation
10. Which of the following statements about carboxylic acids is/are
correct?
1. Simple aliphatic carboxylic acids are soluble in water.
2. Higher carboxylic acids are practically insoluble in water.
3. The acidity of a carboxylic acid depends on the number of carbon atoms
present in the hydrocarbon chain.
Select the correct answer using the code given below.
(a) 1 and 2 only
(b) 2 and 3 only
(c) 1 only
(d) 1, 2 and 3
11. Match List-I with List-II and select the correct answer using the code
given below the Lists :
List - I (Compound) List- II (Type of polymer)
A. Melamine 1. Linear
B. Polyvinyl chloride 2. Condensation
C. Terylene 3. Natural
D. Cellulose 4. Cross-linked
Code :
(a) A B C D
3 2 1 4
(b) A B C D
4 1 2 3
(c) A B C D
3 1 2 4
(d) A B C D
4 2 1 3
12. The Tropic of Cancer passes through which one of the following
countries?
(a) North Sudan
(b) Chad
(c) Mali
(d) Yemen
13. Gersoppa Falls is formed by which one of the following rivers?
(a) Lohit
(b) Tungabhadra
(c) Sharavati
(d) Krishna
14. Which one of the following processes is not a part of long-wave
radiation?
(a) Conduction
(b) Scattering
(c) Convection
(d) Radiation
15. In mid-latitude regions, the diurnal variation in weather is due to
(a) raising air/convection
(b) sinking air/conduction
(c) advection
(d) radiation
16. Springs are common in which of the following areas?
1. Well jointed rocks
2. Arid areas with underlying rocks
3. Karst topography
4. Tilted strata
Select the correct answer using the code given below.
(a) 1 and 3 only
(b) 1, 3 and 4
(c) 2 and 4
(d) 3 and 4 only
17. Consider the following diagram :
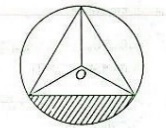
An equilateral triangle is inscribed in a circle of radius 1 unit. The area
of the shaded region, in square unit, is

18. In an examination, 53% students passed in Mathematics, 61% passed in
Physics, 60% passed in Chemistry, 24% passed in Mathematics and Physics, 35% in
Physics and Chemistry, 27% in Mathematics and Chemistry and 5% in none. The
ratio of percentage of passes in Mathematics and Chemistry but not in Physics in
relation to the percentage of passes in Physics and Chemistry but not in
Mathematics is
(a) 7:5
(b) 5:7
(c) 4:5
(d) 5:4
19. Consider the following diagrams:
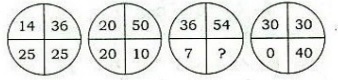
Which one of the following is the missing number in the diagrams given above?
(a) 1
(b) 2
(c) 3
(d) 4
20. Which one of the following is the area of a sector of a circle of
radius 10 cm formed by an arc length of 15 cm?
(a) 107 cm
(b) 158 cm
(c) 75 cm2
(d) 150 cm2
21. On simplification the product
(x1 - y1)(x2 + y2) ... (x10 + y10)
how many such terms are there which will have only single x and rest y's ?
(a) 210
(b) 10
(c) 20
(d) 1
22. Which one of the following statements about blood elements is correct?
(a) Red blood cells' number is same as that of white blood cells and
platelets.
(b) The number of white blood cells is more than red blood cells.
(c) The number of red blood cells is the highest.
(d) The number of red blood cells is lower than platelets.
23. Which one of the following statements regarding muscle contraction and
relaxation is correct?
(a) It involves movement of Zn++ ion.
(b) It involves Ca++ ion of blood.
(c) It involves release of Ca++ from sarcoplasmic reticulum (SR) and pumping
Ca++ back in SR.
(d) It involves exchange of Na and K.
24. Which one of the following statements regarding digestion is correct?
(a) Fat present in the food is digested by trypsin and chymotrypsin.
(b) Starch of food is digested by lipases.
(c) Fat of food is digested by lipases.
(d) Nucleic acid is digested by amylases.
25. Which one of the following statements is correct?
(a) In photosynthesis by green plants, carbon dioxide is used for making
carbohydrate.
(b) During photosynthesis, plants use oxygen for making carbohydrate.
(c) The carbohydrate generated by plants comes from soil.
(d) The carbohydrate generated by plants comes from urea.
26. Which one of the following statements regarding insulin hormone is
correct?
(a) Insulin is a protein hormone which is not produced in human body.
(b) Insulin is a steroid hormone which is produced in human body.
(c) Insulin is a cholesterol-driven hormone given to human.
(d) Insulin is a protein hormone which is produced in human body.
27. Match List-I with List-II and select the correct answer using the code
given below the Lists :
List-I (Limestone region) List-II (Country)
A. Pennines 1. France
B. Yucatan Peninsula 2. Jamaica
C. Cockpit Country 3. Britain
D. Causses 4. Mexico
Code :
(a) A B C D
3 4 2 1
(b) A B C D
1 2 4 3
(c) A B C D
3 2 4 1
(d) A B C D
1 4 2 3
28. Which of the following cities experience the warm temperate
Mediterranean climate?
1. Cape Town
2. Los Angeles
3. Adelaide
4. Santiago
Select the correct answer using the code given below.
(a) 1 and 2 only
(b) 3 and 4 only
(c) 1, 2 and 3 only
(d) 1, 2, 3 and 4
29. Which one of the following is the correct descending order of Indian
States in terms of their density of population (as per 2011 Census)?
(a) Bihar-Kerala-Uttar Pradesh-West Bengal
(b) West Bengal-Bihar-Kerala-Uttar Pradesh
(c) Bihar-West Bengal-Kerala-Uttar Pradesh
(d) Kerala-West Bengal-Bihar-Uttar Pradesh
30. Which one of the following major river basins of India is the shortest
in length and smallest in terms of catchment area?
(a) Cauvery
(b) Mahi
(c) Penner
(d) Sabarmati
31. Arabian Sea records higher salinity than the Bay of Bengal because
(a) Arabian Sea exhibits high rate of evaporation and low influx of fresh
water
(b) Arabian Sea has shallow water
(c) Arabian Sea has more enclosed land
(d) Arabian Sea receives more rainfall
32. At what time between 2 o'clock and 3 o'clock will the hour and minute
hands of a clock be 12 minutes division apart?
(a) 12 minutes past 2 o'clock
(b) 18 minutes past 2 o'clock
(c) 24 minutes past 2 o'clock
(d) 30 minutes past 2 o'clock
33. If the number
22 x 54 x 46 x 108 x 610 x 1512 x 814 x 20+ x 1018 x 2520
is divisible by 10n, then which one of the following is the maximum value
of n?
(a) 78
(b) 85
(c) 89
(d) 98
34. Suppose a, b, c, d and e are five consecutive odd numbers in ascending
order. Consider the following statements :
1. Their average is (a+ 4).
2. Their average is (b+2)
3. Their average is (e-4).
Which of the statements given above is/are correct?
(a) 1 only
(b) 2 and 3 only
(c) 1 and 3 only
(d) 1, 2 and 3
35. A king ordered to make a crown from 8 kg of gold and 2 kg of silver.
The goldsmith took away some amount of gold and replaced it by an equal amount
of silver and the crown when made, weighed 10 kg. The king knows that underwater
gold loses 1/20th of its weight, while silver loses 1/10th. When the crown was
weighed underwater, it was 9.25 kg. How much gold was stolen by the goldsmith?
(a) 1 kg
(b) 2 kg
(c) 3 kg
(d) 4 kg
36. If a cubical container of length, breadth and height each of 10 cm can
contain exactly 1 litre of water, then a spherical container of radius 10.5 cm
can contain
(a) not more than 4 litres of water
(b) more than 4 litres but less than 4-5 litres of water
(c) more than 4-5 litres but less than 5 litres of water
(d) more than 5 litres of water
37. Which one of the following sites does not have coral reef formation?
(a) Gulf of Cambay/Khambhat
(b) Gulf of Mannar
(c) Lakshadweep
(d) Andaman and Nicobar Islands
38. Which of the following statements are appropriate to Mangrove Sites?
1. Mangrove plants require appropriate mix of saline water and freshwater.
2. Mangrove plants require mudflats to enable it to grow and develop.
3. Mangrove plants are found in the inter-tidal zones of sheltered coasts.
4. Mangrove vegetation has been reported in all the coastal States including
Andaman and Nicobar Islands.
Select the correct answer using the code given below.
(a) 1 and 2 only
(b) 2, 3 and 4 only
(c) 1, 3 and 4 only
(d) 1, 2, 3 and 4
39. Which one of the following pairs is not correctly matched?
(a) Indira Gandhi Rashtriya Uran Akademi : Fursatganj
(b) National Flying Training Institute : Belgaum
(c) National Institute of Hydrology : Roorkee
(d) National Water Academy (Headquarters) : Khadakwasla
40. The lowest temperature is observed/recorded in which one of the
following layers of the atmosphere?
(a) Stratosphere
(b) Mesosphere
(c) Thermosphere
(d) Troposphere
41. Which one of the following ocean currents is not a warm current?
(a) North Pacific
(b) Falkland
(c) North Equatorial
(d) Canary
42. What is the largest value for n (natural number) such that 6n divides
the product of the first 100 natural numbers?
(a) 18
(b) 33
(c) 44
(d) 48
43. The angle between the hour hand and the minute hand of a clock at 10
minutes past 3 is
(a) 30°
(b) 35°
(c) 37.5°
(d) 40°
44. An international conference is attended by 65 people. They all speak
at least one of English, French and German language. Suppose 15 speak English
and French, 13 speak English and German, 12 speak French and German and 5 speak
all the three languages. A total of 30 people can speak German and 30 can speak
French. What is the number of people who can speak only English?
(a) 17
(b) 20
(c) 22
(d) 40
45. Suppose 72 = m x n, where m and n are positive integers such that
1<m<n. How many possible values of mare there?
(a) 5
(b) 6
(c) 10
(d) 12
46. Suppose x, y, z are three positive integers such that x<y<z and xyz =
72. Which one of the following values of S yields more than one solution to the
equation x + y - z = S?
(a) 13
(b) 14
(c) 15
(d) 16
47. Which one of the following is the remainder when 1020 is divided by 7?
(a) 1
(b) 2
(c) 4
(d) 6
48. Which one of the following is anisotropic in nature?
(a) Glass
(b) Rubber
(c) Plastic
(d) Quartz
49. Which one of the following is not a condensation polymer?
(a) Nylon
(b) DNA
(c) Polythene
(d) Bakelite
50. The tides whose height is 20 percent more than normal tide are called
(a) spring tides
(b) neap tides
(c) apogean and perigean tides
(d) daily and semi-diurnal tides
51. Which of the following statements about tropical cyclone are correct?
1. It originates and develops over warm oceanic surface.
2. Presence of Coriolis force is necessary for it.
3. It occurs in middle latitudinal region.
4. It develops “eye” with calm and descending air condition.
Select the correct answer using the code given below.
(a) 1 and 2 only
(b) 1, 2 and 4
(c) 3 and 4 only
(d) 2, 3 and 4
52. Which of the following statements about biodiversity hot spots is/are
correct?
1. Biodiversity hot spots are identified by the International Union for
Conservation of Nature and Natural Resources (IUCN).
2. Biodiversity hot spots are defined according to their vegetation.
3. In India, Eastern Ghats and Western Himalaya are the biodiversity hot spots.
Select the correct answer using the code given below.
(a) 1 and 2 only
(b) 3 only
(c) 1, 2 and 3
(d) 1 only
53. Which one of the following is not an igneous rock?
(a) Granite
(b) Gneiss
(c) Pumice
(d) Basalt
54. Which one of the following rivers is west flowing?
(a) Godavari
(b) Periyar
(c) Tungabhadra
(d) Cauvery
55. Consider an equilateral triangle ABC as given in the following diagram
:
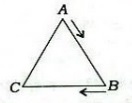
Two people start at the same time from points A and B with speeds 30 km per
hour and 20 km per hour respectively, and move on the sides of the triangle in
the clockwise direction. They meet each other for the first time at
(a) point C
(b) a point between C and A
(c) a point between A and B
(d) point A
56. The number of ways in which 3 boys and 2 girls can be arranged in a
queue, given that the 2 girls have to be next to each other, is
(a) 12
(b) 24
(c) 48
(d) 120
57. The ratio of ages of a man and his son is 3:1. After 15 years, the age
ratio will be 2:1. What is the age of the man?
(a) 45 years
(b) 40 years
(c) 35 years
(d) 30 years
58. Two pillars are placed vertically 8 feet apart. The height difference
of the two pillars is 6 feet. The two ends of a rope of length 15 feet are tied
to the tips of the two pillars. The portion of the length of the taller pillar
that can be brought in contact with the rope without detaching the rope from the
pillars is
(a) less than 6 feet
(b) more than 6 feet but less than 7 feet
(c) more than 7 feet but less than 8 feet
(d) more than 8 feet
Directions : The following eight (8) items consist of two statements,
Statement I and Statement II. Examine these two statements carefully and select
the correct answer using the code given below.
Code :
(a) Both the statements are individually true and Statement II is the correct
explanation of Statement I
(b) Both the statements are individually true but Statement II is not the
correct explanation of Statement I
(c) Statement I is true but Statement II is false
(d) Statement I is false but Statement II is true
59. Statement I:
Sound waves can travel through vacuum.
Statement II :
Light is an electromagnetic wave and can travel through vacuum.
60. Statement I:
A compass needle placed near a current-carrying wire will get deflected.
Statement II :
A current-carrying wire creates magnetic field around it.
61. Statement I:
For early societies, historians retrieve information about elite families
mostly from inscriptions.
Statement II :
Satavahana inscriptions generally end with the term Putra, a Prakrit word
meaning 'son'.
62. Statement I :
In 1856, Nawab Wajid Ali Shah was dethroned and exiled to Calcutta on the
plea that the region was being misgoverned.
Stateinent II :
The Nawab was accused of being unable to control the rebellious chiefs and
Taluqdars.
